Dr. Zahra Zakeri
Professor
Ph.D., St John’s University.
Office: E -114 – Tel: (718) 997 – 3417 Lab: E – 143
Tel: (718) 997- 3429
E-mail: Zahra_zakeri @ hotmail.com
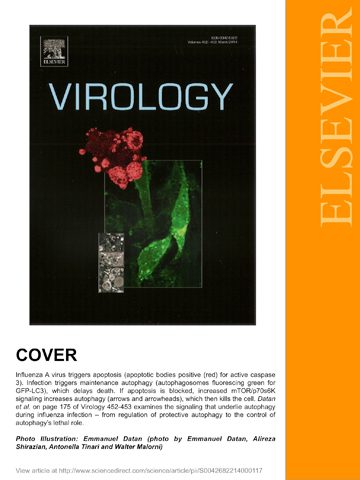
We recently published our work on influenza-induced autophagy and cell death, as the cover story in Virology on Feb 5, 2014. This article can be found in the links below:
Research interests:
Our basic interest is in cellular response mechanisms. Our work has focused primarily on programmed cell death and apoptosis, and more recently on how gender differences affect the fate of the cell and hence the organism.
In the last few years there has been an explosion in the study of how the cells die. Cell death is a fundamental aspect of embryonic development, normal cellular turnover and maintenance of homeostasis. It plays an essential role in diseases such as Acquired Immune Deficiency Syndrome (AIDS), cancer, Alzheimer’s and other degenerative diseases. During development of embryos, cell death shapes and influences the function of almost all organs. In fact, without cell death there would be no embryo. However, too much or too little cell death can be detrimental to the embryo. Alteration of the correct pattern of cell death can result in developmental defects including neural defects, cleft palate, and numerous malformations.
In my laboratory we are studying the mechanisms of cell death. The questions we are asking are, how does a cell know it is to die? What are the specific signals that a cell recognizes that determine its commitment to die? How are these signals transmitted through the cell? And what genes or factors do they affect? What are the specific steps that are initiated to start and continue the demise of the cell? To address these questions we have taken a multidisciplinary approach. We have combined molecular and cellular methodology in vitro with the study of whole animal. We go from looking at the external signals to the identification of activation or deactivation of specific genes and proteins.
We have used developing mouse embryos as well as the metamorphosing tobacco hornworm (Manduca sexta), and Drosophila to examine the normal and abnormal pattern of cell death, In these systems we have examined and characterized different types of cell death, and have identified genes that show specific patterns of expression. In search of mechanisms of cell death, in addition to developing embryos we use a variety of cell lines to investigate signal transduction pathways and second messengers. One such second messenger under investigation in my laboratory is ceramide, the product of the cleavage of cell membrane sphingomyelin by sphingomyelinase. We have discovered that this lipid can act as a second messenger determining the function of neurotrophin receptors.
 One of the main interests of the laboratory is to understand the role of cell death in diseases such as cancer and infection. In these lines we study the regulation of cell cycle genes in cell death and cellular differentiation. We are interested in determining the role of cell cycle genes as well as the effect of chemotherapeutic agents in cell death and during tumorigenesis. We have found that some of the cell cycle genes play an important role in cell death. Using similar approaches to study cell death and cancer we study the pathways involved in neuronal degeneration. Our study of neuronal degeneration has shown the importance of cell cycle and ceramide signaling in cell death. For the study of cell death during infection we have asked how influenza virus kills cells. This study examines the basic mechanism of cell death induction as well as more clinically relevant information, such as how viruses manipulate the machinery of the cell and ultimately activate cell death mechanisms.
One of the main interests of the laboratory is to understand the role of cell death in diseases such as cancer and infection. In these lines we study the regulation of cell cycle genes in cell death and cellular differentiation. We are interested in determining the role of cell cycle genes as well as the effect of chemotherapeutic agents in cell death and during tumorigenesis. We have found that some of the cell cycle genes play an important role in cell death. Using similar approaches to study cell death and cancer we study the pathways involved in neuronal degeneration. Our study of neuronal degeneration has shown the importance of cell cycle and ceramide signaling in cell death. For the study of cell death during infection we have asked how influenza virus kills cells. This study examines the basic mechanism of cell death induction as well as more clinically relevant information, such as how viruses manipulate the machinery of the cell and ultimately activate cell death mechanisms.
Our understanding of regulation of cell death will be important in controlling tumor growth, developmental abnormalities, and neurodegenerative disease, as well as in the treatment of viral infection.
Looking at cell death has led us to examine cellular response to various challenges and has led us to a new question that we are now asking: whether cellular response differs in cells originating from males or females. Our preliminary results indicate that the answer is yes. We are now asking what are the underlying reasons for this gender difference in cellular response andÊ how they relate to the skewing that is seen in acquisition of disease with regard to gender.
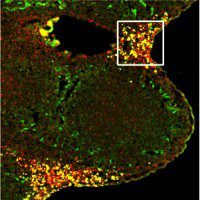
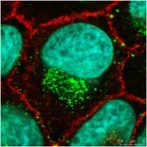 Modoc virus, a flavivirus related to Dengue and West Nile viruses, is shown found in vesicles formed during virus-induced autophagy (green) near the nucleus (blue) of an infected cell (red). Jeff McLean in Dr. Zahra Zakeri’s lab at Queens College has Identified a viral gene responsible for flavivirus-induced autophagy and have characterized its role in the life and death struggle of an infected cell.
Modoc virus, a flavivirus related to Dengue and West Nile viruses, is shown found in vesicles formed during virus-induced autophagy (green) near the nucleus (blue) of an infected cell (red). Jeff McLean in Dr. Zahra Zakeri’s lab at Queens College has Identified a viral gene responsible for flavivirus-induced autophagy and have characterized its role in the life and death struggle of an infected cell.
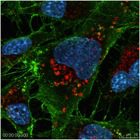 Influenza A virus is shown being assembled in the cytoplasm of infected cells. The virus nucleoprotein (green) attaches to virus RNA in the nucleus (blue) and transports the virus RNA to assembly centers in the cytoplasm where mature viruses are produced. Lysosomes are stained in red. Jeff McLean in Dr. Zakeri’s lab at Queens College has identified a cell death-related protein that controls Influenza virus replication in part by controlling this process.
Influenza A virus is shown being assembled in the cytoplasm of infected cells. The virus nucleoprotein (green) attaches to virus RNA in the nucleus (blue) and transports the virus RNA to assembly centers in the cytoplasm where mature viruses are produced. Lysosomes are stained in red. Jeff McLean in Dr. Zakeri’s lab at Queens College has identified a cell death-related protein that controls Influenza virus replication in part by controlling this process.
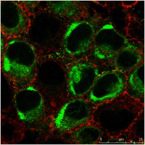 Dengue virus, a deadly tropical flavivirus, is shown in vesicles formed during virus-induced autophagy (green) near the nucleus (blue) of an infected cell (red). Dr. Zahra Zakeri’s lab at Queens College has Identified a viral gene responsible for flavivirus-induced autophagy and have characterized its role in the life and death struggle of an infected cell.
Dengue virus, a deadly tropical flavivirus, is shown in vesicles formed during virus-induced autophagy (green) near the nucleus (blue) of an infected cell (red). Dr. Zahra Zakeri’s lab at Queens College has Identified a viral gene responsible for flavivirus-induced autophagy and have characterized its role in the life and death struggle of an infected cell.
Selected Publications:
1. Singh Ahuja, H., Zhu, Y., and Z. Zakeri. (1997). Association of cyclin-dependent kinase 5 and its activator p35 with apoptotic cell death. Developmental Genetics. 21:258-267.
2. Kagan, T., and Z. Zakeri. (1998). Detection of apoptotic cells in the nervous system. In Harry, J. “Neurodegeneration methods and protocols” in Methods in Molecular Medicine. Vol. 22 Neurodegeneration Methods and Protocols. Humana Press. 105-124.
3. Karasavvas, N., and Z. Zakeri., (1999). Relationships of apoptotic signaling mediated by ceramide and TNF-alpha in U937 cells. Cell Death and Differentiation. 6, 115-123.
4. Nikezic-Ardolic, M., Lin, L., Milcevic, M., and Z. Zakeri. (1999). Gender differences in cellular response. Lupus. 8, 380-383.
5. Lockshin RA, and Z. Zakeri (2001). Programmed cell death and apoptosis: Origins of the theory. Nature Reviews Mol Cell Bio. 2: 544-550.
6. Zhu, Y., L, Lin., S. Woo, D, Quaglino., R, A. Lockshin., and Z. Zakeri (2002). Cyclin Dependent Kinase 5 and Its Interacting Proteins in Cell Death Induced in vivo by Cyclophosphamide in Developing mouse embryos. Cell Death Diff.,9:421-430.
7. (BOOK) Lockshin RA, Zakeri Z, and Tilly JR, eds. (1998). When Cells Die: A Comprehensive Evaluation of Apoptosis and Programmed Cell Death. Wiley-Liss, New York.
8. (BOOK) Lockshin RA, Zakeri Z. (2004). When Cells Die II: A Comprehensive Evaluation of Apoptosis and Programmed Cell Death. Wiley-Liss, New York.
9. L. Lin, C. Penaloza, Y. Ye, R.A. Lockshin, Z. Zakeri, (2009) Detection of Apoptosis in Mammalian Development, in Peter Erhardt and Ambrus Toth, eds., Methods in Molecular Biology (Apoptosis Second Edition), Humana Press, Chapter 18 :pages 259-272.
10. J. E. McLean, E. Datan, D. Matassov, and Z. Zakeri 2009, Lack of Bax Prevents Influenza A Virus-Induced Apoptosis and Causes Diminished Viral Replication. J. Virology, Aug. 2009, 83: 8233-8246
11. Penaloza C., Estevez B., Orlanski S., Sikorska M., Walker R., Smith C., Smith B., Lockshin R.A., Zakeri Z. (2009) Sex of the cell dictates its response: differential gene expression and sensitivity to cell death inducing stress in male and female cells. FASEB J. Jun. 2009 23(6):1869-79
12. Y. Ye, A. Tinari, V. Malorni, R.A. Lockshin and Z. Zakeri, 2009, Activation of Cyclin-Dependent Kinase 5 (Cdk5) is a Consequence of Cell Death. J. Biomedicine Biotechnology in press (available online athttp://www.hindawi.com/journals/jbb/aip.805709.html)
13. Z. Zakeri, N. Maghsudi, F. Motamedi, and R.A. Lockshin 2009, Editorial (Meeting report): “Manipulation of cell pathways in drug development” and introduction to this issue, Gastroenterology and Hepatology from Bed to Bench Vol. 2 #4 (supplement) pp S1-S2 (available online as http://www.ghfbb.ir/index.php/ghfbb/article/view/49/50)